
Symposium for a $175B biopharma company
Keystone organizes workshops that align stakeholders for complex litigations.

Keystone organizes workshops that align stakeholders for complex litigations.
Keystone led a symposium for a $175 billion biopharma and its external counsel. The symposium educated key stakeholders on the complexities of biologics manufacturing and built a knowledge foundation for future cases. Keystone developed the custom curriculum, recruited industry experts and facilitated the workshop.
Keystone workshops follow a process to ensure stakeholders are aligned and educated before going into complex litigation. These educational symposiums are facilitated by Keystone leaders and the course curriculum is developed specifically for the upcoming case or issue. Keystone brings in academic and industry experts who complement and extend Keystone's own expertise to ensure a deeper understanding of technical, scientific, economic, and strategic topics.
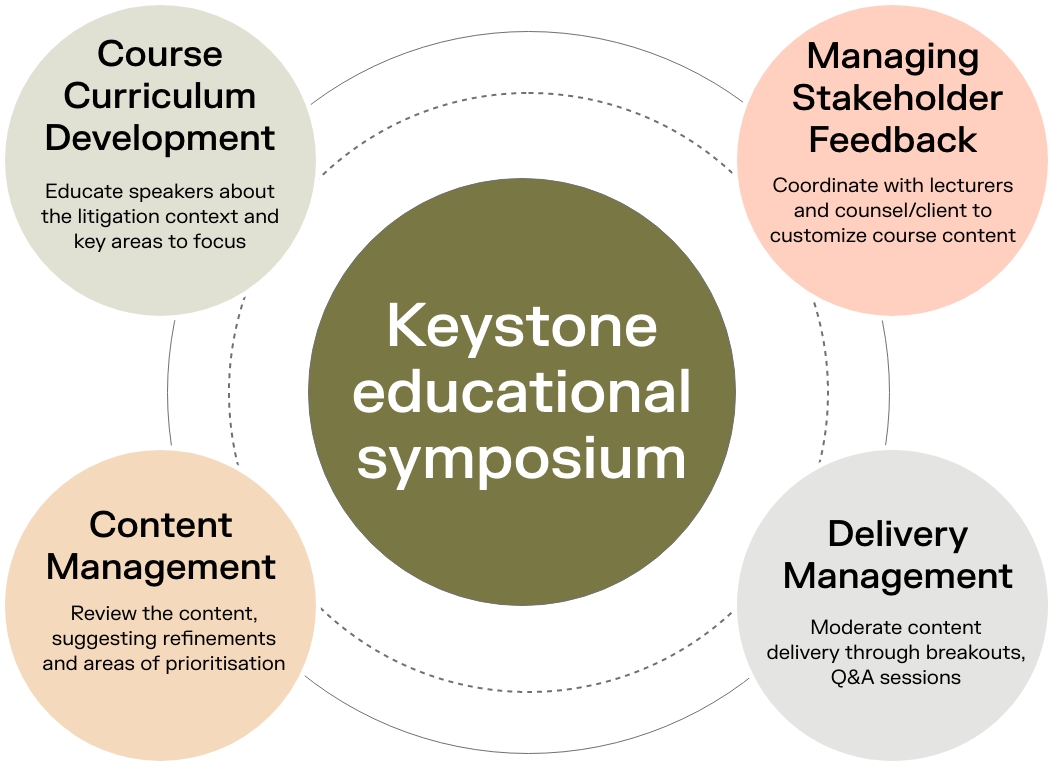
Keystone brought together legal, academic and industry experts to ensure all team members were educated on biologics manufacturing. The workshop helped the legal team be more prepared and fully aligned entering the case.

To learn more about Keystone's approach to industry workshops or our expertise in Healthcare and Life Sciences litigation services and strategy, contact science@keystone.com.